Therefore atoms that are present on one side of the equation must be present on the other side as well. Fe o 2 fe 2o 3.
Count your atoms on each side.

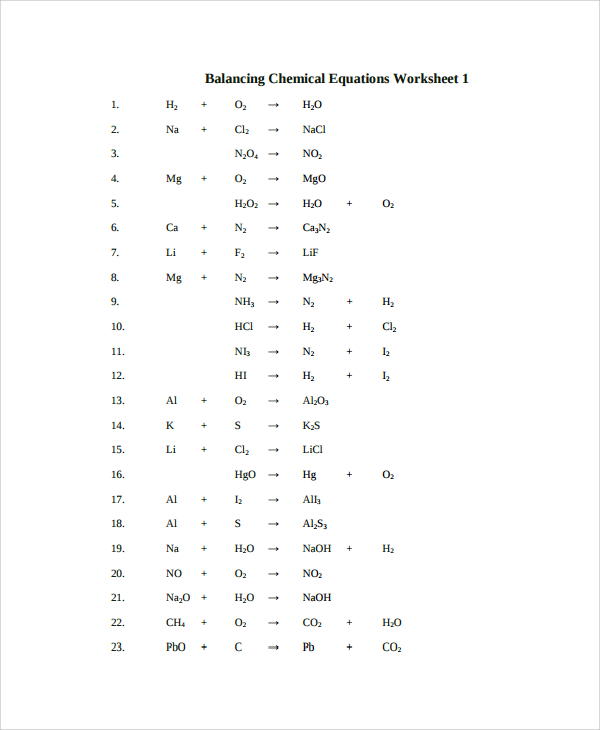
How to balance chemical equations for beginners pdf.
Find suitable coefficients which are the numbers placed before chemical formulas to indicate how many units of each substance are required to balance the equation.
Aloh3 h2so4 al2so43 h2o counting the atoms of each element in the equation it is found that there are 1 atom al 7 atoms o 5 atoms h and 1 atom s on the left side and 2 atoms al 13 atoms o 2 atoms h and 3 atoms s on the right side.
This equation would have also balanced with coefficients of 2 6 and 4 instead of 1 3 and 2.
To do this keep in mind a subscript indicates the number of atoms.
Reactant product this equation is unbalanced.
H2 o2 h2o.
A balanced chemical equation gives the number and type of atoms participating in a reaction the reactants products and direction of the reaction.
Balancing an unbalanced equation is mostly a matter of making certain mass and charge are balanced on the reactants and products side of the reaction arrow.
By no means should you add subscripts because this will completely change the formula of the particular reactant or compound causing a change in the entire meaning that the equation wants to render.
The equation is balanced.
Then add coefficients to the atoms on each side of the equation to balance them with the same atoms on the other side.
Another example of balancing an equation is.
Keep organized and make a table underneath the reactants and products with the number of elements involved in the reaction.
1 nitrogen molecule reacts with 3 hydrogen molecules to yield 2 ammonia molecules.
You can read the equation this way.
To balance a chemical equation start by writing down the number of atoms in each element which is listed in the subscript next to each atom.
The next step for balancing the chemical equation is to determine how many atoms of each element are present on each side of the arrow.
Balanced only with coefficients while balancing the chemical equations balance them only by placing coefficients in front of them.
Be balanced on both sides according to the law of conservation of mass which says that matter cannot be created or destroyed.
For example o 2 has 2 atoms of oxygen.
















No comments:
Post a Comment